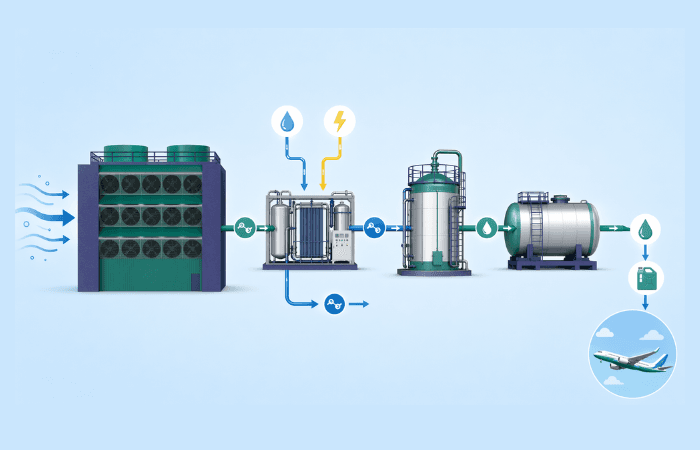
Hydrogen Electrolysers Co-Located with Direct Air Capture for eFuel Production
23 April 2026
Locating a Direct Air Capture (DAC) unit alongside a green hydrogen electrolyser is an elegant way to produce low-carbon synthetic fuels.
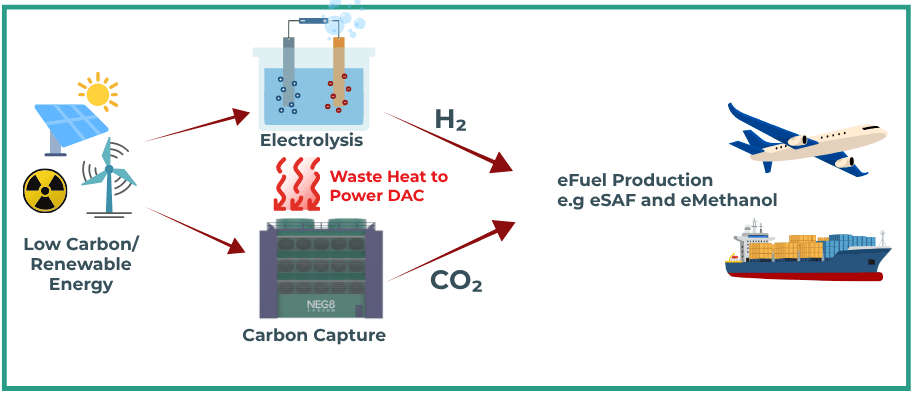
The reason is simple. The electrolyser supplies green hydrogen (H2), while the DAC unit supplies carbon dioxide (CO2) captured from the ambient air. The H2 and the CO2 can then be made into synthetic fuels such as eMethanol and, through further upgrading, eKerosene (electro Sustainable Aviation Fuel or eSAF) and other synthetic hydrocarbons.
When the electricity comes from renewable or near-zero carbon generation to make green hydrogen and to power certain DAC processes, and when the DAC unit uses low-temperature heat efficiently, the overall system makes technical and commercial sense.
Using DAC with Hydrogen Electrolysers for eFuels Production
The DAC-plus-electrolyser concept is often presented as a route not just to one fuel, but to a family of eFuels.
An integrated eFuel production site would include four linked sections:
- A low-carbon power supply
- A hydrogen electrolyser
- A low-temperature DAC unit
- A fuel synthesis plant
The first step is hydrogen production.
Water electrolysis is already commercially available through alkaline and PEM systems, which are currently the main options for projects linked to variable renewable power.
The second step is carbon capture.
In a DAC system, a sorbent selectively binds CO2 from the atmosphere. Once the sorbent is saturated, the CO2 is released again in concentrated form. (See: How does Direct Air Capture Work?) This regeneration step is where heat demand becomes important. Solid sorbent DAC is attractive here as it typically needs low-grade heat, often in the range of about 80 to 120°C, with NEG8 Carbon regenerating at a low 65°C.
The third step is synthesis.
The simplest reaction is methanol synthesis from carbon dioxide and hydrogen:
CO2 + 3H2 -> CH3OH + H2O
Methanol is useful in its own right, but it is also a platform molecule that can be converted into other fuels and chemicals. Methanol is the most straightforward first product because it sits at the simplest end of the carbon-to-fuel chain. It only needs CO2 and H2 and not the extra process complexity required for longer-chain hydrocarbon fuels. It can be directly used in shipping (eMethanol), and it can also act as an intermediate for more complex synthetic fuels like eKerosene/eSAF or Methanol-to-Jet Fuel. In other words, methanol is both a fuel and a building block.

eSAF for Aviation
Aviation needs a hydrocarbon fuel with tighter performance requirements. So, the methanol is upgraded further in a chain of operations to produce eKerosene/eSAF (Sustainable Aviation Fuel). In one route, methanol is produced first and then converted into jet-range hydrocarbons. In another, CO2 is converted via reverse water-gas shift into a CO-rich gas mixture, which is then processed through Fischer-Tropsch synthesis and upgrading. Either way, the hydrogen still comes from electrolysis and the carbon can come from DAC. Both routes are under active development for power-to-liquid (PtL) Sustainable Aviation Fuel.
That is why co-location is essential. It does not remove the complexity of fuel synthesis, but it improves the starting conditions as it gives the plant direct access to hydrogen, direct access to captured CO2 and an avenue to reuse heat that would otherwise be wasted.
Importance of Low-Carbon Electricity and Waste Heat in eFuel Manufacturing
Even with good heat integration, the dominant energy input to this chain remains electricity, especially for hydrogen production. If the electricity is fossil-based, the emissions benefit falls away, which is why low-emission eFuels require low-emission power.
The IEA states that eFuels are low-emission only when the hydrogen is made with low-emission electricity and when the carbon input is sourced in a way that keeps life-cycle emissions low. This definition also includes nuclear power which can provide near-zero carbon electricity at a high capacity factor which is good for plants that need steady operation.
For solid sorbent DAC, the regeneration step is one of the main energy demands. If that heat has to be generated separately with electric heaters or dedicated boilers, cost rises. On the other hand, if it can be drawn from a nearby source of low-grade heat, the economics improve. This is where the electrolyser becomes more than a hydrogen unit, it also becomes a heat source.
PEM and alkaline electrolysers reject low-grade heat, commonly around 60-90°C, which is a good match to the 65-120°C heat demand of solid-sorbent DAC. A 2023 study on waste heat utilisation from PEM electrolysers found in their case the electrolyser worked on average at a 75% load and 80% efficiency (HHV), so the theoretical waste heat potential of the electrolyser stack would be 20% of the electrical input, with ~14% usable heat (electrolyser operating at 65 °C). [van der Roest et al., Utilisation of waste heat from PEM electrolysers]. That recovered heat does not remove the need for electricity, but it reduces the external heat duty that the DAC plant would otherwise need to buy or generate.
Solid oxide electrolysers operate at a much higher temperature, about 700-800°C, so any recoverable heat is higher grade.
| Electrolyser Type | Technology | Waste Heat / Heat Figure | Potential Use for DAC |
| Proton Exchange Membrane (PEM) | Solid polymer membrane conducts protons. | Operates at 70-90°C. 14-15% of stack electricity input could be used by a heat consumer. | Good fit for solid DAC, but heat upgrading may be needed if below 80°C. |
| Alkaline (AWE) | Liquid alkaline electrolyte, usually KOH, conducts hydroxide ions. | Commercial alkaline units are typically below 100°C. A 100 MW case study modelled extractable heat at 80 °C. | Good fit for solid DAC at low-temperature regeneration. |
| Solid Oxide (SOEC) | Ceramic electrolyte conducts oxygen ions using steam at high temperature. | Operates at about 700-800°C | Could supply higher-temperature DAC heat, but heat is often reused internally first. |
[Source: US Department of Energy]
USING THE FISCHER-TROPSCH PROCESS HEAT
There is also heat recovery possible from the Fischer-Tropsch process used to produce eFuel, which ranges from 3.1 to 5.2 MWh per tonne of CO2 utilised.
Increased Efficiency and Lower Costs
Overall, the gain is that the combined site becomes more thermally integrated. Heat that would have been rejected is instead used to help regenerate the DAC sorbent. This raises plant efficiency and can lower operating cost, and that is the real value of co-location.
To sum up, the sources of heat for DAC from eFuel production:
- Electrolyser Heat (major constituent at 52-59%)
- Syngas & Syncrude Cooling
- Flue Gas
- Fischer Tropsch Flue Gas
Solid Sorbent DAC Works Well for Integration with Hydrogen Electrolysers
For this type of integrated plant, solid sorbent DAC is the best fit owing to the low temperature level needed for regeneration and the ability to use indirect heat. These solid sorbent DAC systems can use low-grade heat, whereas high-temperature solvent-based approaches have a poorer temperature match with electrolyser waste heat. (See: Solid Sorbent DAC Compared with Electrochemical DAC)
Decarbonisation and CO2 Utilisation
Capturing CO2 is part of broader decarbonisation efforts and the captured CO2 can also be used in other processes or permanently stored underground if it’s not used to make eFuels.
Conclusion
Direct Air Capture and hydrogen electrolysis fit together well in making carbon-based eFuels. The pairing works because each unit supplies what the other part of the fuel chain needs. The electrolyser supplies hydrogen and the DAC plant supplies CO2. Most importantly, a low-temperature solid sorbent DAC process can also make use of the electrolyser’s low-grade waste heat, which improves the thermal fit of the whole plant and lowers operating cost.